Hygiene precautions and, especially in the case of many viruses, vaccinations are essential measures to prevent infectious diseases. In addition, bacterial films on surgical equipment, but also in food production, must be prevented or regularly removed.
Infections – Prevention
Sterilization of medical equipment with plasma and UV technology

In traditional sterilization methods, microorganisms are inactivated through various physical or chemical agents such as heat, radiation or the influence of gases. The choice of the sterilization method depends on the material composition, the objective of the treatment and the range of application. With thermolabile materials such as medical products or equipment in medical engineering, thermal sterilization processes are not suitable. Gas sterilization procedures using explosive, toxic or carcinogenic gases are not optimal as well because they require high safety standards.
The sterilizing action of low-temperature plasmas – inactivating microbial cells – offers a material-friendly alternative. In addition, the reactive particles in the plasma (excited molecules, radicals, ions accelerated in the electric field, and photons) remove organic contaminations such as pyrogenic cell residues. The sterilizing gas mixtures are in fact produced directly in the plasma and no costly disposal is necessary.
In addition, scientists at Fraunhofer IGB have further developed UV treatment to minimize the number of reproductive microorganisms on surfaces. With new, specially designed excimer lamps or the latest UV LED technology, packaging foils, but also filling systems in the pharmaceutical industry or medical engineering surfaces can be sterilized effectively and quickly.
The processes can be scaled up and adapted to individual requirements. Fraunhofer IGB has many years of experience in the field of plasma and UV sterilization. In addition to determining the microbicidal effectiveness of different radiation/plasma sources, the institute is engaged in the optimization and further development of such systems.
On-site production of hydrogen peroxide for disinfectants
Due to its highly oxidizing and cell toxic effects hydrogen peroxide is widely used as a bleaching agent and disinfectant in many areas, in particular as a basic agent of disinfactants. Mainly, hydrogen peroxide is produced in large scale using the anthraquinone process. Disinfectants on the basis of hydrogen peroxide are supplied to the users as ready-made solutions. However, in the event of disruptions in production or supply bottlenecks due to external influences such as the current pandemic crisis, an adequate supply of this disinfectant cannot be guaranteed. In hygienically and medically sensitive situations such as the current pandemic crisis, an inadequate supply of disinfectants however becomes a serious problem for hospitals, for example.

One solution to this challenge is offered by the production of hydrogen peroxide solutions and their specific formulation as disinfectants, directly on site. Fraunhofer IGB has developed and implemented a new technical concept where hydrogen peroxide is generated in an electrochemical cell, that means with just (air) oxygen, water and electricity. The core component of this cell is a gas diffusion electrode (GDE) at which the production of hydrogen peroxide from gaseous oxygen and water takes place in a targeted manner.
Current work focuses on the further development of the electrolytic cell and the process control in order to generate the required concentrations of hydrogen peroxide directly by means of electrosynthesis and to integrate it into a plant concept including a dosing system. The next goal is a prototype that can be tested by potential users on site and with which consumers can produce their disinfectants fully automatically and independently of external conditions.
Vaccine development
New inactivation method for killed vaccines
Inactivated vaccines, so called dead vaccines, are composed of pathogens that are usually inactivated using toxic chemicals such as formaldehyde. This process is tedious and the chemicals have to be removed again from the preparation. In addition, important protein antigens are often destroyed by the chemical inactivation making the vaccine less effective.
In the Fraunhofer ELVIRA project, the four Fraunhofer institutes IZI, IGB, FEP and IPA took an interdisciplinary approach to develop a physical method that successfully overcomes these problems. Viruses, bacteria, or parasites can now be inactivated in seconds – instead of days or even weeks – and without the addition of toxic substances such as formaldehyde using low-energy electron radiation. At the same time, important antigens in the vaccine are preserved, while the nucleic acids of the viruses and bacteria are reliably destroyed by the electrons.
In a follow-up project funded by the Bill and Melinda Gates Foundation, the researchers have developed two prototypes for the automatic inactivation of polio pathogens.
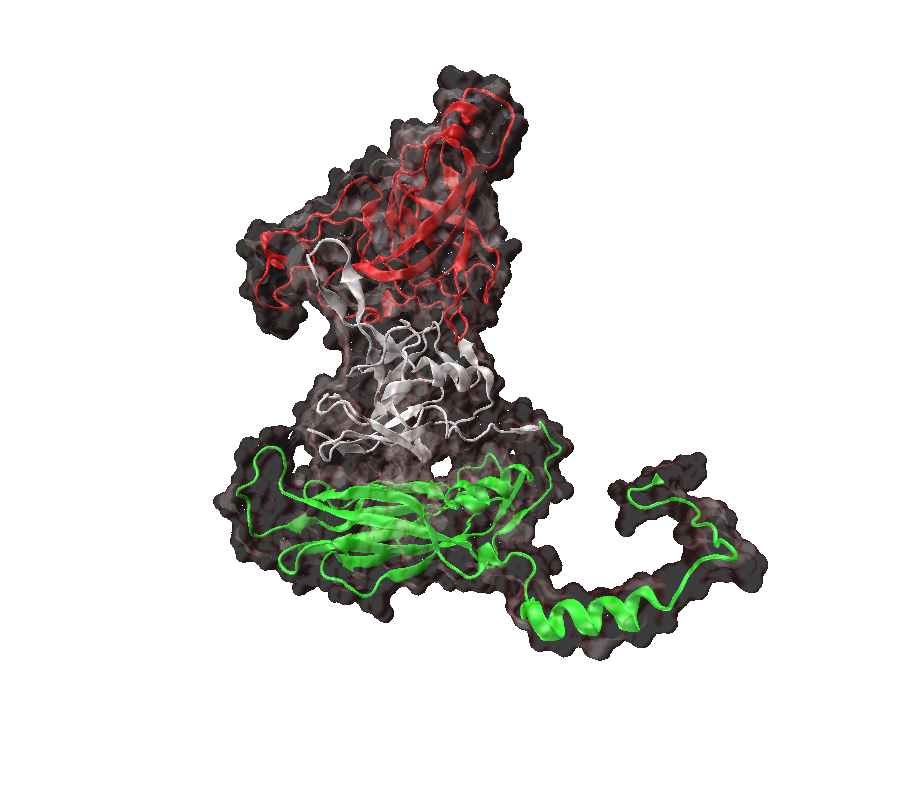
Engineering platform for vaccines
The dramatic development of the corona epidemic with worldwide effects requires quick solutions, especially in the areas of prevention and therapy. Vaccinations in particular are effective measures to prevent recurring epidemics and to protect people against infectious diseases.
The use of platform technologies to facilitate adaptation is essential for the rapid availability of vaccines against emerging pathogens.
At Fraunhofer IGB a virus engineering platform has been developed which can be used as a basis for therapeutic purposes (oncolytic viruses), but also for prevention (vaccines). By incorporating an appropriate vaccine antigen into the virus platform, carrier viruses can be produced as potential vaccines. The modular viral system can also be quickly adapted in the future in response to newly emerging pathogens and as a combination vaccine, thus providing significant support in coping with pandemics.
Virus-like particles – Biocontainers for vaccination and drug delivery
Virus-like particles (VLP) are biobased capsules that mimic viruses and can be used in various ways. Due to their stability, size and multivalence, VLPs are excellently suited as a basis for vaccines, for example against viruses that cannot or are difficult to grow in vitro, or against foreign proteins that are presented on their surface. They are also suitable for packaging and drug delivery, as they can transport therapeutic agents in high concentrations, with few side effects and specific targeting.
So far, the broad use of VLPs is limited, however, by the lack of standardized and efficient platform technologies for their production, which would allow different VLPs to be brought to different sites of action with different loads, similar to a modular system. The Fraunhofer Vari-VLP project has successfully developed a modular platform technology for the production of VLPs which, with further functional optimization, can be used as VLPs for drug delivery and vaccination.
Biofilms – Characterization, avoidance, removal
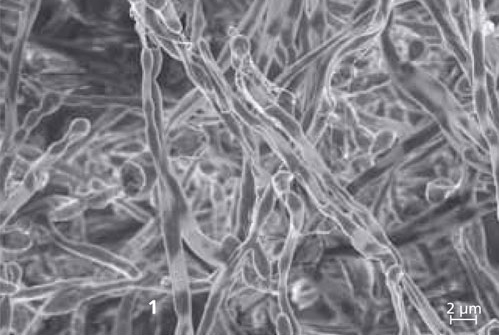
Biofilms are communities of bacteria, fungi or algae that attach themselves to surfaces and grow there – in the body, for example in lung tissue, but also on medical instruments or food processing equipment. They are surrounded by an extracellular matrix, the so-called EPS (extracellular polymeric substances), which is formed by the bacteria themselves. The EPS consists mainly of polysaccharides, but also contains a complex mixture of proteins, DNA and lipids. This shields the microorganisms from the environment like a protective cover, for example attacks of the immune system or chemical substances such as antibiotics or disinfectants.
For several years, Fraunhofer IGB has been working on questions in which the interactions between microbial cells and surfaces or the formation of biofilms play a role. For example, we have developed test procedures for the examination of biofilms and examine surfaces and components in medical engineering and hygiene for microbiological contamination.
Antimicrobial finishing of surfaces
In order to prevent the development of biofilms, the application of suitable antimicrobial surface modifications, for example by binding biocidal substances or photocatalytic equipment, is indicated. An appropriate surface design can prevent microbial adhesion to the material surface or cell proliferation from the outset. In cooperation of interfacial engineering researchers with microbiologists and cell biologists we have already developed various surface finishes at the IGB. Physical and chemical methods are available for characterization, and we use cell-based systems to prove biological effectiveness.
Bacteriophages against biofilms
Bacteriophages are viruses that specifically and exclusively destroy bacteria. In Eastern Europe, phages have been successfully used for decades for the treatment of bacterial inflammation, such as burns, inflammation of the bone marrow and retina or cystic fibrosis-associated infections. In particular, they represent an alternative to antibiotics that is increasingly coming into focus.

In addition, phages are suitable wherever contaminating bacteria need to be specifically removed, for example for food preservation or for the degradation of biofilms in medical engineering. Fraunhofer IGB is also working on the use of bacteriophages for targeted reduction of germs. The key points here are the identification, engineering, cultivation, storage and formulation of phages. With Kari-EX, a Fraunhofer funded project, a novel, cost-effective and safe method for the causal control of caries has been developed using phages.
 Fraunhofer Institute for Interfacial Engineering and Biotechnology IGB
Fraunhofer Institute for Interfacial Engineering and Biotechnology IGB