Biomaterials for treatment of demyelination disorders of the central nervous system
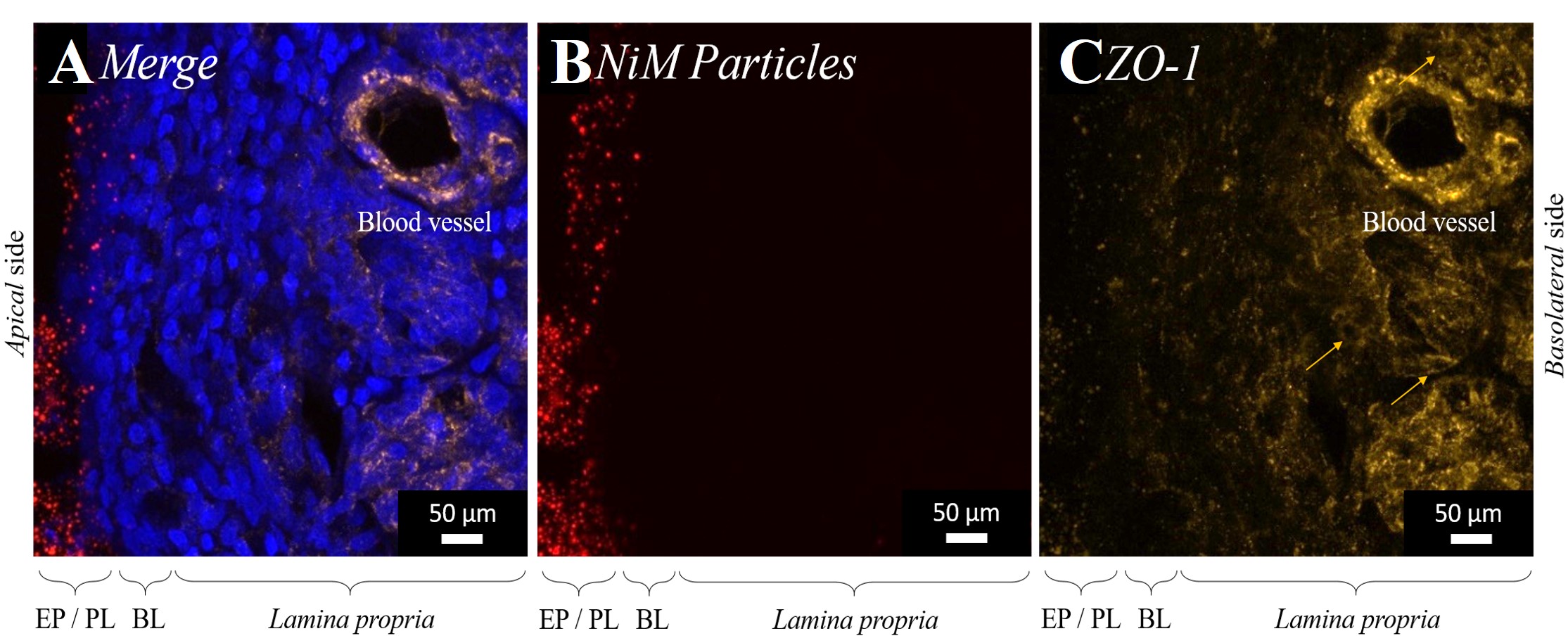
Effective drugs for the treatment of diseases of the central nervous system (CNS) exist. However, the blood-brain barrier (BBB), which protects the brain as the body's control center, makes it difficult for therapeutic biomolecules in particular to pass through. This can be remedied by nose-to-brain transport via the olfactory nerve, which enables the direct passage of therapeutic antibodies into the CNS. In the N2B-patch project coordinated by Fraunhofer IGB, an intranasal application platform for CNS-active biopharmaceuticals has been developed for this purpose. To do so, an innovative galenical formulation based on biomaterials has been combined with a special applicator and applied as a hydrogel bandage on the olfactory epithelium.

 Fraunhofer Institute for Interfacial Engineering and Biotechnology IGB
Fraunhofer Institute for Interfacial Engineering and Biotechnology IGB